 Discovery Information Discovery Information
|
| Who: Johann Arfvedson |
| When: 1817 |
| Where: Sweden |
|
 Name Origin Name Origin
|
| Greek: lithos (stone). |
| "Lithium" in different languages. |
|
 Sources Sources
|
| Lithium is widely distributed but does not occur in nature in its free form. Because of its reactivity, it is always found
bound with one or more other elements or compounds. Found in trace amounts in the minerals; spodumene (LiAl(SiO3)2), amblygonite (Li,Na)AlPO4(F,OH)), lepidolite (KLi2Al(Al,Si)3O10(F,OH)2). Most commercial lithium is recovered from brines sources in Chile. Also obtained by passing electric charge through melted lithium chloride.
|
| Around 39 thousand tons are produced every year. |
|
 Abundance Abundance
|
| Universe: 0.006 ppm (by weight) |
| Sun: 0.00006 ppm (by weight) |
| Carbonaceous meteorite: 1.7 ppm |
| Earth's Crust: 20 ppm |
| Seawater: 0.18 ppm |
| Human: |
| 30 ppb by weight |
| 27 ppb by atoms |
|
 Uses Uses
|
| Used in batteries, ceramics, glass, lubricants, alloy hardeners, pharmaceuticals, hydrogenating agents, heat transfer liquids, rocket propellants, vitamin A synthesis, nuclear
reactor coolant, underwater buoyancy devices and the production of tritium. Deoxidizer in copper and copper alloys.
|
| Alloys of the metal with aluminium, cadmium, copper, and manganese are used to make high performance aircraft parts.
|
| Lithium salts such as lithium carbonate (Li2CO3), lithium citrate, and lithium orotate are mood stabilizers (the effect is due to the lithium ion, so the type of salt is unimportant). They are used in the treatment of bipolar disorder, since unlike most other mood altering
drugs, they counteract both mania and depression. Lithium can also be used to augment other antidepressant drugs. Useful amounts
of lithium for this use are only slightly lower than toxic amounts, so the blood levels of lithium must be carefully monitored
during such treatment.
|
| Lithium metal is used as a catalyst in some types of methamphetamine production, particularly in illegal amateur "meth labs."
|
|
 History History
|
| Petalite (LiAlSi4O10), which has lithium in it, was discovered by the Brazilian scientist Jose Bonifacio de Andrade e Silva in the late 1700s on a trip to Sweden. Lithium was discovered by Johan August Arfwedson in 1817. Arfwedson found the new element within the minerals spodumene
and lepidolite in a petalite ore (LiAl(Si2O5)2) that he was analyzing during a routine investigation of some minerals from a mine on the island Uto in Sweden. In 1818 Christian Gmelin was the first to observe that lithium salts give a bright red colour in flame. Both men tried and
failed to isolate the element from its salts.
|
| The element was not isolated until William Thomas Brande and Sir Humphry Davy later used electrolysis on lithium oxide in
1818. Robert Bunsen and Matiessen isolated larger quantities of the metal by electrolysis of lithium chloride in 1855. Commercial
production of lithium metal was achieved in 1923 by the German company Metallgesellschaft through using electrolysis of molten
lithium chloride and potassium chloride. It was apparently given the name "lithium" because it was discovered from a mineral
while other common alkali metals were first discovered from plant tissue.
|
|
 Notes Notes
|
| Lithium is a soft, silvery metal, so soft that it can be cut with a sharp knife. It is the lightest of all metals and has
a density only half that of water.
|
| Lithium is one of only three elements - and the only metal - created in the first moments of the Big Bang. (The other two
elements are hydrogen and helium, which according to cosmologists, were created in much greater abundance than lithium.)
|
|
 Hazards Hazards
|
| Lithium causes serious burns, especially when in contact with damp skin. Contact with the eyes may cause serious permanent
damage.
|
| It is the only metal that reacts with nitrogen at room temperature. Near its melting point, lithium ignites in air. Lithium posses a dangerous fire and explosion risk when exposed to water, acids or oxidizing agents. Lithium fires are difficult to extinguish, requiring special chemicals designed to smother them. It
reacts exothermally with nitrogen in moist air at high temperatures. In solution lithium is toxic and targets the central nervous system.
|




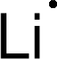
 Discovery Information
Discovery Information
 Name Origin
Name Origin
 Sources
Sources
 Abundance
Abundance
 Uses
Uses
 History
History
 Notes
Notes
 Hazards
Hazards
 Images
Images

