| Reactions of Europium |
 Reactions with water Reactions with water |
| Europium is quite electropositive and reacts slowly with cold water and fairly quickly with hot water to form europium hydroxide,
and hydrogen gas.
|
2Eu(s) + 6H2O(g)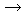 2Eu(OH)3(aq) + 3H2(g) 2Eu(OH)3(aq) + 3H2(g) |
 Reactions with air Reactions with air |
| Europium metal burns readily in air to form europium(III) oxide. |
4Eu + 3O2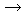 2Eu2O3 2Eu2O3 |
 Reactions with halogens Reactions with halogens |
| Europium metal reacts with all the halogens to form europium(III) halides. |
2Eu(s) + 3F2(g)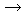 2EuF3(s) 2EuF3(s) |
2Eu(s) + 3Cl2(g)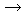 2EuCl3(s) 2EuCl3(s) |
2Eu(s) + 3Br2(g)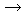 2EuBr3(s) 2EuBr3(s) |
2Eu(s) + 3I2(g)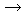 2EuI3(s) 2EuI3(s) |
 Reactions with acids Reactions with acids |
| Europium metal dissolves readily in dilute sulphuric acid to form solutions containing the very pale pink Eu(III) ion together with hydrogen gas.
|
2Eu(s) + 3H2SO4(aq)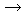 2Eu3+(aq) + 3SO42-(aq) + 3H2(g) 2Eu3+(aq) + 3SO42-(aq) + 3H2(g) |




 Reactions with water
Reactions with water 2Eu(OH)3(aq) + 3H2(g)
2Eu(OH)3(aq) + 3H2(g) Reactions with air
Reactions with air 2Eu2O3
2Eu2O3 Reactions with halogens
Reactions with halogens 2EuF3(s)
2EuF3(s) 2EuCl3(s)
2EuCl3(s) 2EuBr3(s)
2EuBr3(s) 2EuI3(s)
2EuI3(s) Reactions with acids
Reactions with acids 2Eu3+(aq) + 3SO42-(aq) + 3H2(g)
2Eu3+(aq) + 3SO42-(aq) + 3H2(g)